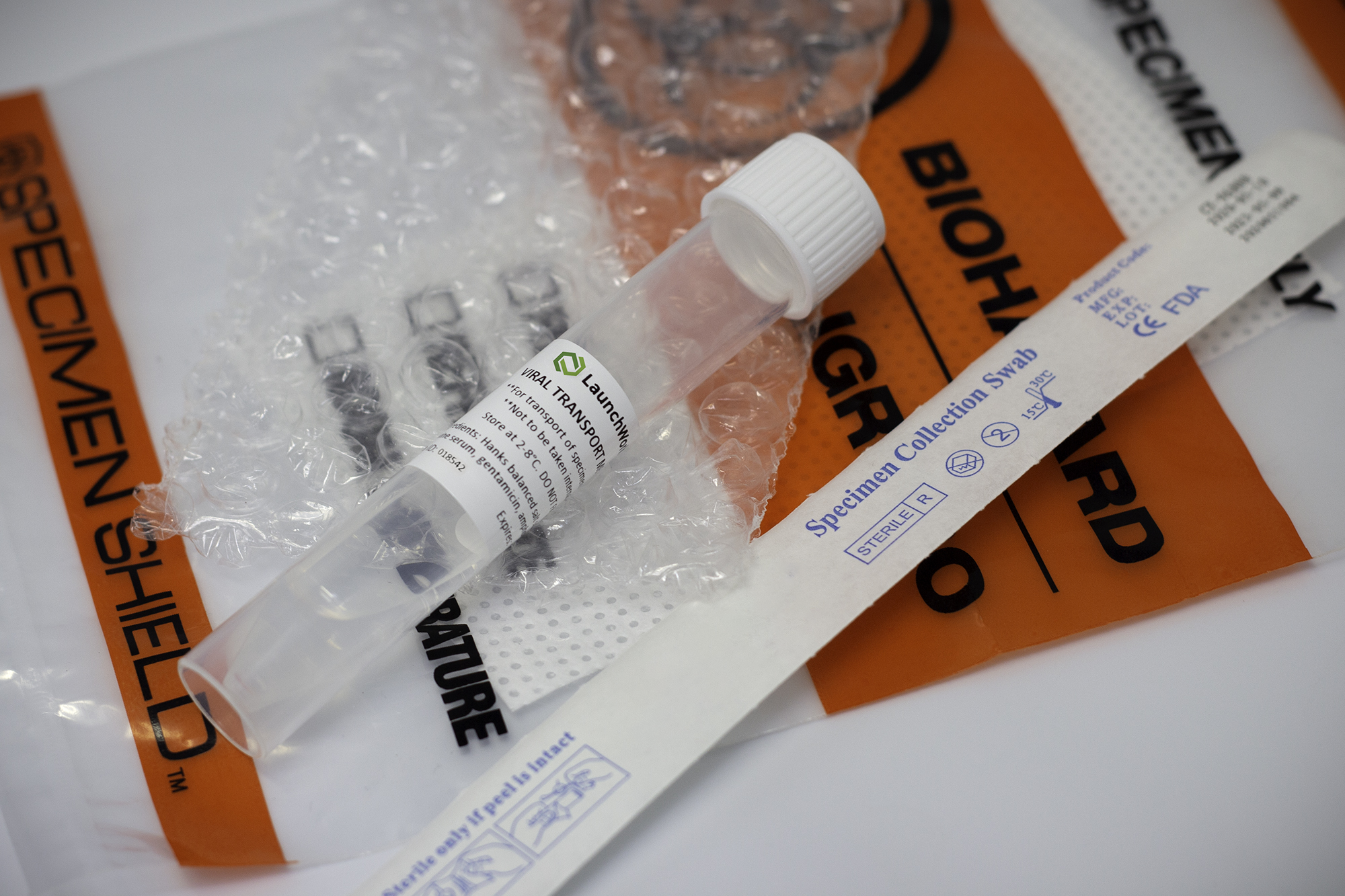
LaunchworksCDMO added to FDA list of approved vendors for validating and distributing VTM, in accordance with COVID-19 EUA Transport Media Policy
LaunchWorksCDMO, a contract development and manufacturing company serving the life science market, has been approved to manufacture and distribute Viral Transport Media, in accordance with Section IV.B of the FDA’s COVID-19 Transport Media Policy.. The company’s Viral Transport Media is available in the form bulk tubes, large volume containers and a specimen kit, which will include a tube of sterile LaunchWorks VTM, a biohazard bag and an absorbent bag. Additional options include Nylon-flocked NP swab, Barcode or QP-coded labels, patient card, and a UN 3373 return box.
The Viral Transport Media is a new addition to the LaunchWorks suite of products and service offerings. As an FDA-registered ISO 13485 certified company, LaunchWorks is a division of Integreon Global, which has supported LaunchWorks’ expansion in molecular diagnostics with the additions of new service offerings, including embarking on an automation program designed to help its customers be more competitive. Recently, a significant portion of the company’s efforts have involved the response to the COVID-19 crisis, and the development of Viral Transfer Media is a valuable addition.
“We’re grateful to contribute to the fight against the pandemic, and to add this important component to our product portfolio,” says Jeff Kelly, General Manager of LaunchWorks CDMO. “Our team has worked hard to bring this product to market, and the addition brings us one step closer to offering an end-to-end solution for our customers supporting the fight against COVID-19.
”This reinforces the LaunchWorks’ commitment to continue to expand and support the world’s changing needs due to the pandemic, he continued. “The Launchworks service suite ranges from package design to formulation, filling and fulfillment, making us uniquely positioned to help companies translate their science into commercial products. We take pride in our ability to contribute to the resulting improvement in world health.”
LaunchWorks VTM and other COVID-19 information can be viewed here.
###
About LaunchWorks
LaunchWorks is a winning team of innovative, experienced, and knowledgeable contract manufacturing experts with a track record of turning complex life science ideas into high-quality products on-time and on-budget. They are committed to accelerating life science product commercialization by optimizing every step of the manufacturing process in order to create high-quality products for our customers that reach the market and the people that need it, safely and quickly. For more information visit www.launchworkscdmo.com
About Integreon Global
Founded in 1990, Integreon is a complete packaging solutions provider dedicated to providing goods and services to maintain the integrity of our clients’ products. Our team includes a full complement of engineers, designers, and manufacturing specialists with the collective skills, gathered in one close-knit organization, to assure easy access to leading experts. For more information visit www.integreonglobal.com